|
11/14/2023 0 Comments Definition of sigma bondThe σ orbital (solid color) is filled, while the π* orbital (grayed) is an unpopulated antibonding orbital. Hyperconjugation: orbital overlap between a σ orbital and π* orbital stabilizes alkyl-substituted alkenes. The Baker–Nathan effect, sometimes used synonymously for hyperconjugation, is a specific application of it to certain chemical reactions or types of structures. However, extended versions of hyperconjugation (such as double hyperconjugation ) can be important as well. Only electrons in bonds that are in the β position can have this sort of direct stabilizing effect - donating from a sigma bond on an atom to an orbital in another atom directly attached to it. In particular, the new orbital with bonding character is stabilized, resulting in an overall stabilization of the molecule. Increased electron delocalization associated with hyperconjugation increases the stability of the system. However, sometimes, low-lying antibonding σ* orbitals may also interact with filled orbitals of lone pair character (n) in what is termed negative hyperconjugation. C–H or C–C) with an adjacent unpopulated non-bonding p or antibonding σ* or π* orbitals to give a pair of extended molecular orbitals. Usually, hyperconjugation involves the interaction of the electrons in a sigma (σ) orbital (e.g.  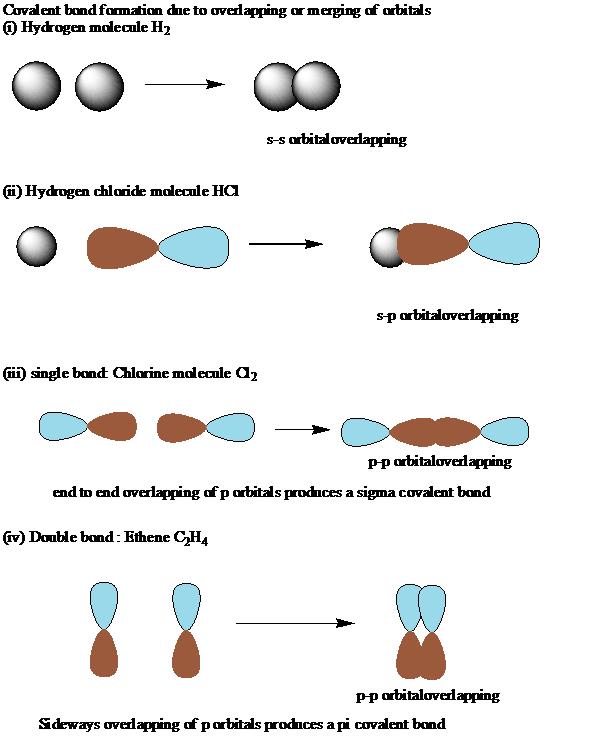
In organic chemistry, hyperconjugation ( σ-conjugation or no-bond resonance) refers to the delocalization of electrons with the participation of bonds of primarily σ-character.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
